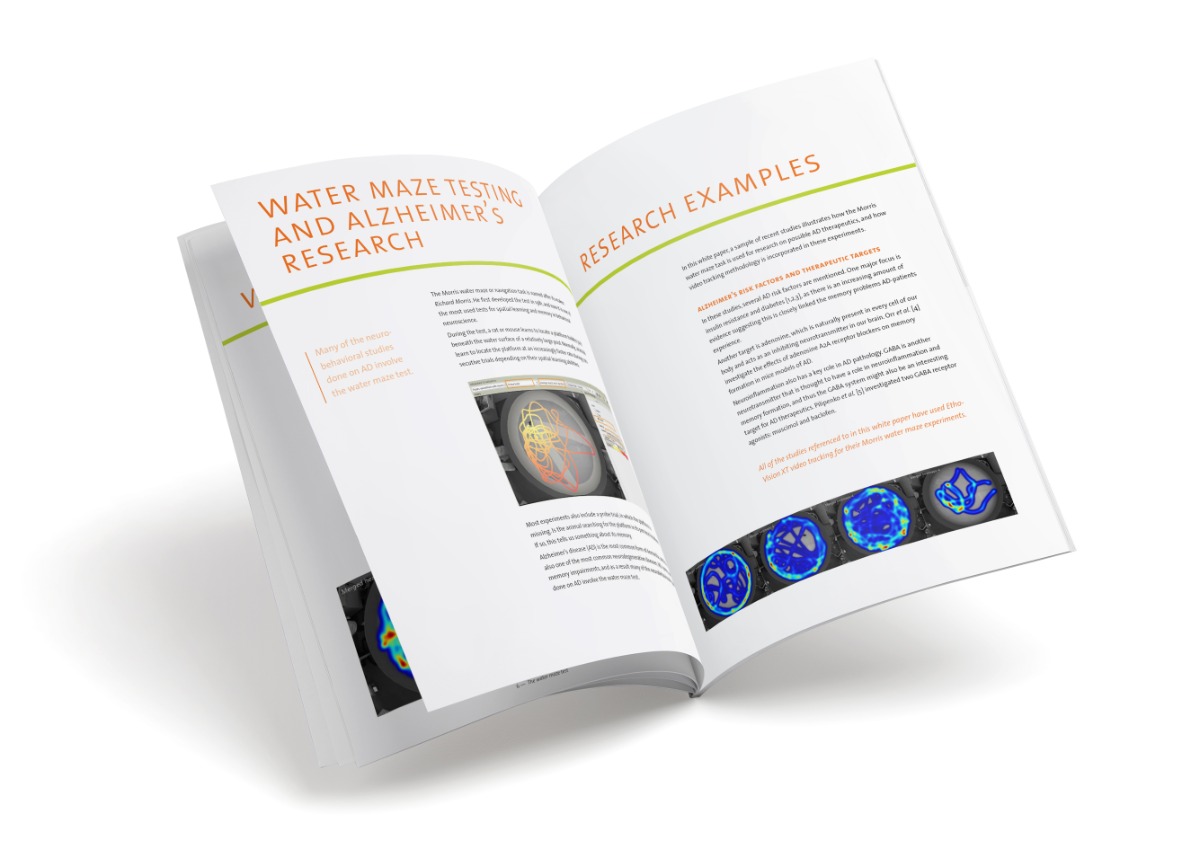
How zebrafish and optogenetics are great for investigating stress
By this point, we do not need to tell you how popular zebrafish are in scientific research. We also probably do not need to point out the great technological advances that are being made in research because of the use of optogenetics. It is safe to say that zebrafish and optogenetics are popular in neuroscience research. Today, let us tell you about the study of De Marco et al. (2013), which combined the two.
Stress in humans and animals
Stress can have detrimental impact on our health and well-being – ranging from cardiovascular effects to depression and other psychiatric disorders. Glucocorticoids (GCs) are the final effector hormones of the brain stress center (hypothalamic-pituitary-adrenal [HPA] axis) and the focus of many animal and human studies.
Investigating glucocorticoids
Traditionally, methods to investigate the effects of GCs include stressor exposure or GC infusion. The problem with using stressor exposure is that it only increases the GC levels via the stimulation of other hormones, since GC is the end-hormone in a cascade of hormone stimulation and release. Direct GC infusion can cause bias because the procedure can be stressful in itself.
Investigating stress with optogenetics
Optogenetics might just hold the answer to the downfalls of traditional methods, because it can offer direct and selective manipulation of GC levels. In fact, De Marco and colleagues effectively developed an optogenetic methods that manipulated the gain of the stress axis with the high temporal accuracy.
Zebrafish as the perfect candidates
The stress response is strongly conserved across species, and in fact, zebrafish have a hypothalamic-pituitary-interrenal axis that is similar to our HPA axis. This is one reason for using zebrafish as a model for stress studies. Here is another: zebrafish larvae are transparent. This makes them great candidates for optogenetic stimulation. Contrary to rodent studies, there is no need for implantation of fiberoptics.
For this study, zebrafish embryos were genetically manipulated to express the light-activated bPAC enzyme in pituitary cells (which govern GC secretion). These embryos were called bPAC+ zebrafish, and compared to their bPAC- siblings in a series of tests.
Light as stressor and stimulator
Of course, optogenetics works with light, and light in itself is a stressor for zebrafish. (Studies show that wild-type larvae show a drastic reduction in locomotor activity in response to light exposure and an increase in locomotion after the light has been turned off, which gradually decreases to normal levels in minutes.) In this study, De Marco and colleagues took advantage of this double-function of light: it is both a stressor that has an indirect influence on GC levels and an optogenetic stimulator with direct influence on GC levels.
Measuring the effects on behavior
To assess the behavioral effects of both direct and indirect manipulation of GC levels, the locomotor activity of 30 zebrafish larvae was measured simultaneously with video tracking software EthoVision XT.

Increase in activity and cortisol levels in response to light
The researchers aimed to increase levels of cortisol (the most important GC) in response to light stimulation. And in fact, bPAC- larvae showed the normal increase in cortisol levels after exposure to blue light, while the bPAC+ larvae showed an even higher increase. It also led to a higher increase in locomotor activity in bPAC+ larvae compared to their negative siblings.
Additionally, early blue-light stimulation caused long term effects; overexposure of GC lead to increased basal cortisol levels in bPAC+ larvae. These larvae also responded to light with higher cortisol levels than bPAC+ larvae that were not exposed to light early on.
Future implications
This study shows a promising method for stress research, as optogenetics seems to provide a way to establish both rapid and delayed changes in the GC-system, detectable both in brain function as well as behavior. For more details on this study, please find the article at http://www.ncbi.nlm.nih.gov/pubmed/23653595.
Read more
- De Marco, R.J.; Groneberg, A.H.; Yeh, C.-M.; Castillo Ramirez, L.A.; Ryu, S. (2013). Optogenetic elevation of endogenous glucocorticoid level in larval zebrafish. Frontiers in Neural Circuits, 7, article 82.
- Read more blog posts on optogenetics
- Read more blog posts on zebrafish
- Read more about zebrafish behavioral research solutions
- Read more about optogenetic solutions for behavioral research
Get the latest blog posts delivered to your inbox - every 15th of the month
more
Getting robust results: one zebrafish is not like the other
Zebrafish larvae locomotor behavior often has a high variability, which can have a big impact on your results. Still, it is one of the most used parameters. So how can you make your study more robust?
How young zebrafish cope with stress
Stress is a natural thing, and how we cope with it differs from person to person. In research, we use the term coping style, something that emerges early on in life for zebrafish.