The response of oxytocin to mother-infant interaction
Researcher Rebecca Knapp was interested to know whether infant eye gaze away from the mother, or eye gaze directed to the mother, would correspond to increased maternal oxytocin. Read her blog post to learn more.
Posted by
Published on
Tue 08 Aug. 2017
Topics
| Eye Contact | Parent-child Interaction | The Observer XT | Video Observation |

Posted by Rebecca Knapp (Deakin University, Centre for Social and Early Emotional Development, Melbourne, Australia).
Background to the research field
Oxytocin
Oxytocin is a hormone with special properties. Everyone produces oxytocin; men, women and children. Originally found to be of particular importance in birth and lactation, researchers have started to look at other, broader influences oxytocin may have on human behavior and relationships. Animal models have found that oxytocin plays a role in a wide range of maternal behaviors and in the quality of maternal caregiving.1
Studies are now looking at whether oxytocin might share a relationship with human maternal and infant behavior as well.1-5
Oxytocin and maternal behavior
In human studies, oxytocin has been found to correspond to increased gaze, touch and positive affect in mothers towards their own infants. In adults, oxytocin has been found to increase trust and to improve the reading of emotional cues; important qualities in a developing relationship.2, 6
The general view of those researching oxytocin’s potential role in maternal and infant relationships suggest that oxytocin supports mutual reward and synchrony. Mothers and infants who share similar oxytocin response to interaction appear to share greater synchrony in those interactions, such as greater mutual gaze, and greater mutual positive affect.7-9
There is some data to suggest that certain maternal behaviors release oxytocin in mothers, and that the release of oxytocin increases certain maternal behaviors in what is considered by some to be a ‘bio-behavioral feedback loop’.10 There is further research to suggest that maternal oxytocin and behavior might influence infant oxytocin and behavior, suggesting that this bio behavioral feedback loop goes beyond the individual, and is expressed between individuals in the mother-infant dyad.

But relationships are not one-way. Mothers respond to infants, and infants respond to mothers. A mother might tickle her baby who shows pleasure by giggling and reaching for more, perpetuating the mother’s behavior. An infant might look away when he has had enough, and the mother waits for her baby’s gaze to return before starting a new game. A baby might shift her gaze to an interesting toy, and when she experiences a moment of delight, looks back to her mother to share this moment.
It is a delicate dance between mother and infant, each responding to, and influencing each other. Part of this dance is one of parental sensitivity and the development of infant attachment security to the caregiver.
Maternal behavior and infant attachment security
In the field of infant attachment research, two maternal behaviors are fundamental to the development of infant attachment security: the mother’s capacity to allow her baby to explore, and her capacity to welcome her infant back to her in times of need.11, 12
Whilst some studies have speculated that oxytocin might support maternal caregiving behavior and thereby support the development of infant attachment security, none have explored specific infant behaviors that might influence maternal oxytocin response and maternal behavior. Synchrony is not enough, as it does not explain maternal readiness to welcome an infant back following a rupture in the relationship or at times of infant need.
Eye gaze, mother-infant communication and oxytocin
Eye gaze between two individuals is central to communication and is a key feature of both maternal sensitivity and infant attachment security.13-17 Gaze aversion is one way infants help regulate their own emotional state, particularly when they feel over-excited.13-17 Infants may also look away as part of their exploration as they take in their surrounding environment.
A mother who experiences her infant’s attentional shift away as a sign of the infant rejecting her will feel and respond differently from a mother who experiences this same behavior as a sign of the infant’s fatigue, boredom or interest in other things. This study sought to investigate whether oxytocin might pay a role in facilitating the openness of the mother to the infant ‘coming back in’ after a break in interaction.
For the purpose of this study, a ‘break in interaction’ was defined as the period during which the infant looked away from his or her mother. I was interested to know whether infant eye gaze away from the mother, or eye gaze directed to the mother, would correspond to increased maternal oxytocin.
If it is synchrony and social reward alone that corresponds to oxytocin release, then one would expect greater infant gaze to the mother to correspond to greater oxytocin concentration in the mother. If oxytocin however serves a function of maintaining maternal engagement through increasing eye gaze, and readiness to welcome the infant back in, then infant disengagement (looking away) should correspond to a maternal oxytocin response.
How the data was gathered
Method
Ten mothers and their infants attended the lab at Deakin University, at a time which fell 1 hour either side of a feed, as salivary oxytocin has been found to peak in that period.18
Following a 10-min familiarization period where mother and infant became accustomed to the room, the mother provided a baseline saliva sample using a Salivette (Sarstedt, Rommelsdorft, Germany).
The mother was then asked to place her baby on an infant beanbag positioned on the floor, and to sit on the floor facing her baby. Two cameras were placed in the room, one facing the mother, the other facing the infant. A third camera behind a two-way screen was positioned to capture the mother and infant in profile.
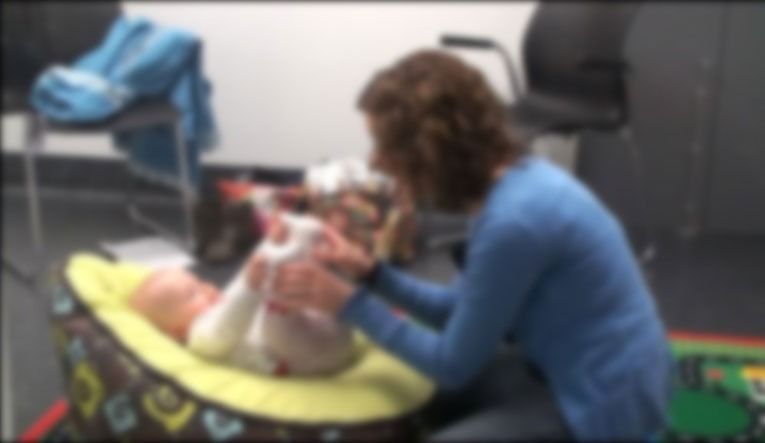
(Image obscured to protect participant privacy)
The three videos were then synchronized and combined into a single split screen image using The Observer XT.
Mothers were asked to engage in 10 minutes of play with their infants without toys. Play could include any type of interaction, game, vocalisation, or touch they typically used. Mothers were told they could pick up their baby if the baby became distressed however they were otherwise encouraged to keep their infant in the beanbag to aid videoing of infant and maternal gaze direction.
Fifteen minutes after the end of the interaction task, a second saliva sample was collected from mothers.
Saliva samples were analyzed by an EIA commercial kit, consistent with previous research.4, 10, 18
Coding of gaze
Gaze was coded separately for mothers and infants, using The Observer XT. Maternal gaze was coded in relation to it being directed to the infant’s face (on/off). Infant gaze was coded in relation to it being directed to the mother’s face (on/off).
Results
First, maternal gaze and maternal oxytocin were explored to see if previous research would be replicated, which it was.
Increasing maternal oxytocin from pre- to post-interaction was associated with increased maternal eye gaze during the interaction task, whilst decreasing maternal oxytocin was associated with decreasing maternal eye gaze.
Then, the relationship between infant eye gaze and maternal oxytocin was explored.
Infants whose gaze increased over the interaction task were compared with infants whose gaze decreased over time. For these two groups, increasing infant gaze was associated with decreasing maternal oxytocin, whilst decreasing infant gaze was associated with a slight increase in maternal oxytocin.
Conclusions
As expected, increasing maternal gaze was associated with increasing maternal oxytocin, consistent with previous research.19 Interestingly, in regards to infant gaze and maternal oxytocin, increasing infant gaze was associated with decreasing maternal OT.
It is possible that when there is greater degree of engagement by an infant with their mother, maternal oxytocin may decrease as the socially affiliative properties of oxytocin are not required to maintain or repair a connection, however when infants are disengaging, maternal oxytocin may be elicited as a social stress response, in order to support maternal readiness for reconnection.
This would be consistent with the observed increase in maternal OT predicting increasing maternal eye gaze, as mothers possibly reorient to and attempt to reengage with their disengaging infants.
Thus, infant gaze away from the mother may elicit a maternal oxytocin response. Oxytocin may serve to increase maternal vigilance of the infant’s emotional state as the infant explores, maintaining the balance between supporting infant exploration, and readiness for her infant to return to the relationship.
Future directions
Of course this was a very small pilot study and these results would need to be replicated on a larger scale to demonstrate reproducibility. Further, it is likely that different mothers responded differently to their infants looking to and away from them, in terms of their own behaviour, their interpretation of their infants’ behaviour, and potentially in terms of their oxytocin response.
It would be interesting in future, larger scale studies to compare two groups – one where infants increased their gaze and the other where infants decreased their gaze over the interaction task, and compare mothers whose oxytocin rose, to those whose oxytocin fell over the course of these two conditions.
Seeing how those mothers responded in terms of their own eye gaze would help determine whether infant behaviour does indeed influence maternal behaviour, and whether maternal oxytocin mediates the relationship between the two.
Thanks to Noldus
I wanted to thank Noldus for very kindly offering an extended trial loan of the Observer XT. The detailed behavioural coding that was required would not have been possible without sophisticated software like The Observer XT. It enabled me to code not only gaze, but several other behavioral variables, quickly, accurately and efficiently. I cannot thank Noldus enough for their generosity, support and their interest in my research project.
References
- Ross, H. E., & Young, L. J. (2009). Oxytocin and the neural mechanisms regulating social cognition and affiliative behavior. Frontiers in Neuroendocrinology, 30(4), 534-547. doi:http://doi.org/10.1016/j.yfrne.2009.05.004
- Feldman, R. (2012). Oxytocin and social affiliation in humans. Hormones and Behavior, 61(3), 380-391. doi:10.1016/j.yhbeh.2012.01.008
- Carter, C. S. (2014). Oxytocin pathways and the evolution of human behavior. Annual Review of Psychology, 65(1), 17-39. doi:10.1146/annurev-psych-010213-115110
- Carter, S. C., Pournajafi-Nazarloo, H., Kramer, K. M., Ziegler, T. E., White-Traut, R., Bello, D., & Schwertz, D. (2007). Oxytocin: behavioral associations and potential as a salivary biomarker. Annals of the New York Academy of Sciences, 1098(1), 312-322. doi:10.1196/annals.1384.006
- Feldman, R. (in press). The neurobiology of mammalian parenting and the biosocial context of human caregiving. Hormones and Behavior. doi:http://dx.doi.org/10.1016/j.yhbeh.2015.10.001
- Lee, H.-J., Macbeth, A. H., Pagani, J. H., & Scott Young 3rd, W. (2009). Oxytocin: The great facilitator of life. Progress in Neurobiology, 88(2), 127-151. doi:http://doi.org/10.1016/j.pneurobio.2009.04.001
- Feldman, R. (2003). Infant-mother and infant-father synchrony: The coregulation of positive arousal. Infant Mental Health Journal, 24(1), 1-23. doi:10.1002/imhj.10041
- Feldman, R. (2007). Parent-infant synchrony and the construction of shared timing; physiological precursors, developmental outcomes, and risk conditions. Journal of Child Psychology and Psychiatry and Allied Disciplines, 48(3-4), 329-354. doi:10.1111/j.1469-7610.2006.01701.x
- Feldman, R. (2012). Interactive synchrony: A biobehavioral model of mutual influences in the formation of affiliative bonds in healthy and pathological development. Neuropsychiatrie de l'Enfance et de l'Adolescence, 60(5), S2. doi:10.1016/j.neurenf.2012.04.016
- Feldman, R., Gordon, I., Schneiderman, I., Weisman, O., & Zagoory-Sharon, O. (2010). Natural variations in maternal and paternal care are associated with systematic changes in oxytocin following parent-infant contact. Psychoneuroendocrinology, 35(8), 1133-1141. doi:10.1016/j.psyneuen.2010.01.013
- Ainsworth, M. D., Blehar, M. C., Waters, E., & Wall, S. (1978). Patterns of attachment: Assessed in the strange situation and at home: Hillsdale, NJ: Erlbaum.
- Marvin, R., Cooper, G., Hoffman, K., & Powell, B. (2002). The Circle of Security project: Attachment-based intervention with caregiver-pre-school child dyads. Attachment & Human Development, 4(1), 107-124. doi:10.1080/14616730252982491
- Beebe, B., Jaffe, J., Markese, S., Buck, K., Chen, H., Cohen, P., Bahrick, L., Andrews, H. & Feldstein, S. (2010). The origins of 12-month attachment: a microanalysis of 4-month mother-infant interaction. Attachment & Human Development, 12(1-2), 3-141. doi:10.1080/14616730903338985
- Beebe, B., Lachmann, F., Markese, S., Buck, K. A., Bahrick, L. E., Chen, H., Cohen, P., Andrews, H., Feldstein, S. & Jaffe, J. (2012). On the Origins of Disorganized Attachment and Internal Working Models: Paper II. An Empirical Microanalysis of 4-Month Mother-Infant Interaction. Psychoanalytic Dialogues, 22(3), 352-374.
- Beebe, B., & Steele, M. (2013). How does microanalysis of mother-infant communication inform maternal sensitivity and infant attachment? Attachment & Human Development, 15(5-6), 583-602. doi:10.1080/14616734.2013.841050
- Beebe, B., Steele, M., Jaffe, J., Buck, K. A., Chen, H., Cohen, P., Kaitz, M., Markese, S., Andrews, H., Margolis, A. & Feldstein, S. (2011). Maternal anxiety symptoms and mother–infant self‐and interactive contingency. Infant Mental Health Journal, 32(2), 174-206.
- Koulomzin, M., Beebe, B., Anderson, S., Jaffe, J., Feldstein, S., & Crown, C. (2002). Infant gaze, head, face and self-touch at 4 months differentiate secure vs. avoidant attachment at 1 year: A microanalytic approach. Attachment and Human Development, 4(1), 3-24. doi:10.1080/14616730210123120
- White-Traut, R., Watanabe, K., Pournajafi-Nazarloo, H., Schwertz, D., Bell, A., & Carter, C. S. (2009). Detection of salivary oxytocin levels in lactating women. Developmental Psychobiology, 51(4), 367-373. doi:10.1002/dev.20376
- Kim, S., Fonagy, P., Koos, O., Dorsett, K., & Strathearn, L. (2014). Maternal oxytocin response predicts mother-to-infant gaze. Brain Research, 1580, 133-142. doi:10.1016/j.brainres.2013.10.050
Related Posts

How to study social behavior and dyadic interactions

