A detailed look at fish larvae
DanioScope
If you are interested in a scope of zebrafish embryo and larvae parameters such as embryo activity, cardiovascular measurements, morphology, and gut and blood flow, then DanioScope is just what you need.
- Automated detection of zebrafish embryos (in chorion) – minimizing manual interference
- Can handle situations in which the animal slightly moves by combining multiple measures on the animal at the different locations
- Acquire data in a batch from list of videos
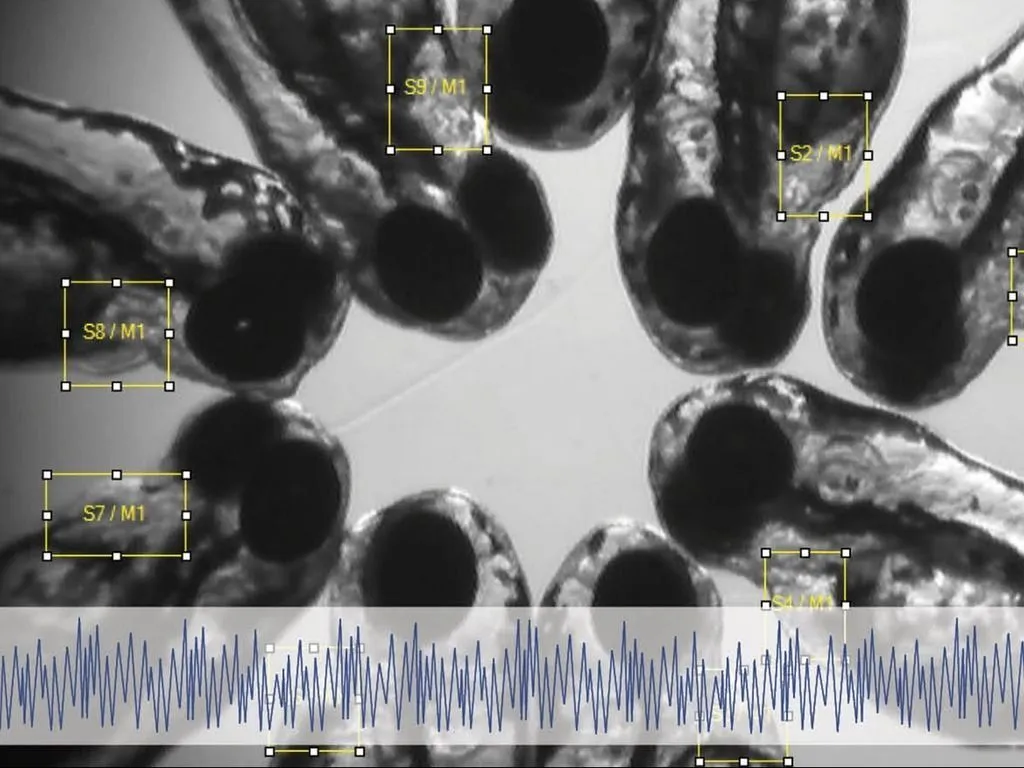
What is DanioScope?
Parameters you can measure
DanioScope non-invasively measures these behaviors – simply use a video of one or more zebrafish embryos. DanioScope automatically recognizes each embryo (within their chorion). Then it measures their activity. If floating occurs you can simply adjust the location of the measurements during the experiment. DanioScope reports back the following parameters:
- Burst activity - percentage of time (from total measurement duration) the embryo was moving
- Inactivity – percentage of time of inactivity (100% - burst activity).
- Burst duration – total time spent active (sum of all movement durations)
- Inactivity duration – total time spent inactive
- Burst count – number of times the embryo moved
- Burst count / per minute – number of movements per minute
The zebrafish cardiac system is assessed by measuring the heart rate or inter-beat interval. Traditional methods include manual counting (using a stopwatch), using a micro pressure system, Laser Doppler microscopy, or electrocardiograms. These methods are labor-intensive, time-consuming, and require specific training to perform.
Video based assessment of heart rate and inter-beat intervals is far less labor intensive. After indicating the heart area in the video imported in DanioScope, it automatically calculates the number of beats per second (BPS) and beats per minute (BPM).
Blood flow can be measured using hand counting methods that often require labor-intensive and time-consuming fluorescence labeling techniques. DanioScope uses video analysis to measure the flow in a blood vessel. In addition, it can measure gut flow in a similar fashion.
To measure flow, you select (a part of) the blood vessel or the part of the gut, from wall to wall, in your video image. Changes in pixels on a frame-by-frame basis is used to measure flow. The blood or gut flow is presented as an activity percentage.
With intuitive drawing tools you can define distances or areas you are interested in, and after calibration DanioScope will measure lengths and surfaces automatically. This way you can easily determine tail length, eye size, and pericardial area. Or any other measurement, because you can define your own!
With images from different time point, you can easily monitor growth and malformations over time.
The features of DanioScope
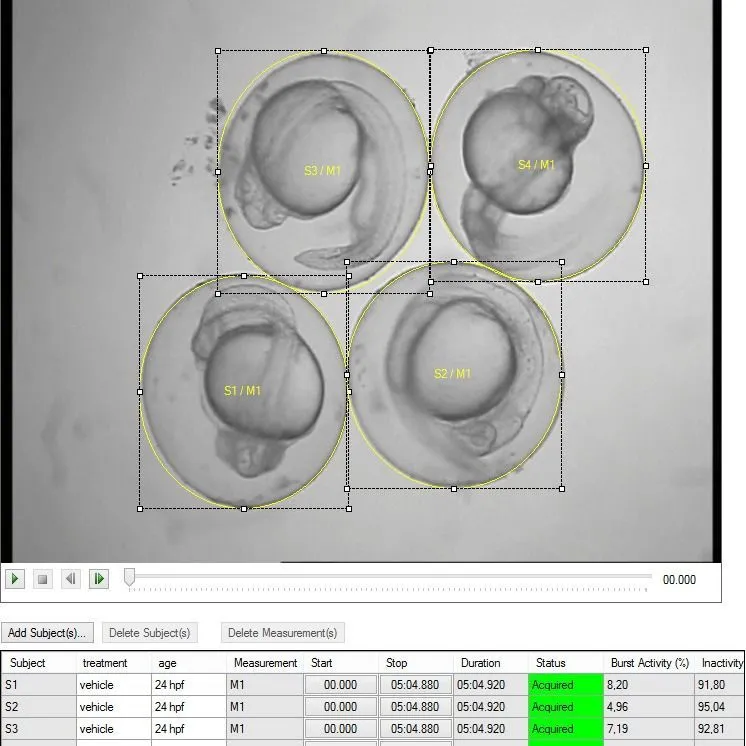
After data acquisition, DanioScope offers options for data selection,
visualization, and analysis. You can also easily export raw data for further analysis in your
statistical program of choice. DanioScope is suited to support high-speed videos (over 60 fps), and
can handle a wide range of video formats.
The software has many features that will make your setting up and executing your experiment easier:
- Combine multiple measures
- Batch acquisition
- Easy experiment handling and organization
- Flexible selecting, grouping and displaying of data
- Allows for the redefining of thresholds to fine-tune results
All I need to know about DanioScope
DanioScope has been published many times over the past years. Because it is an all-in-one tool, DanioScope has been used for many different applications. You can find an overview of recent publication on our resources page.
Ready to take the next step?
We’re passionate about helping you achieve your research goals. Let’s discuss your project and find the tools that fit.
Contact us