Can caffeine prevent Alzheimer's?
A Morris water maze memory study
What is the most popular drug in the world? It’s not alcohol, cannabis, or cocaine, but something most of us start with each day. Coffee; or, more specifically: caffeine. Like millions of other people, it helps me get started and prevents my morning headaches. Caffeine also has been shown to prevent age-related cognitive decline by reducing the risk of developing Alzheimer’s Disease (AD) and dementia. It is not surprising, then, that much research has already been done on the effect of caffeine in the development of AD.
Mouse models of Alzheimer’s disease
AD has two major pathological hallmarks: so-called plaques and tangles in the brain. Amyloid beta peptides accumulate to form plagues outside of the cells, and hyperphosphorylated tau proteins form tangles inside of the cells. Both types of abnormal protein deposits are associated with the cognitive impairments observed with AD. To investigate the effect of caffeine on AD, studies have been done on AD mouse models with amyloid plaques, but previously no work had been done on models based on the tau-tangles. Cyril Laurent and his colleagues thought it was time to change this.
Testing the memory with a water maze
A transgenic mouse model with progressive AD-like tau pathology (THY_Tau22) was tested against a wild-type group. Half of each group received caffeine in their drinking water, resulting in four treatment groups. To test learning and memory, researchers used the Morris water maze.
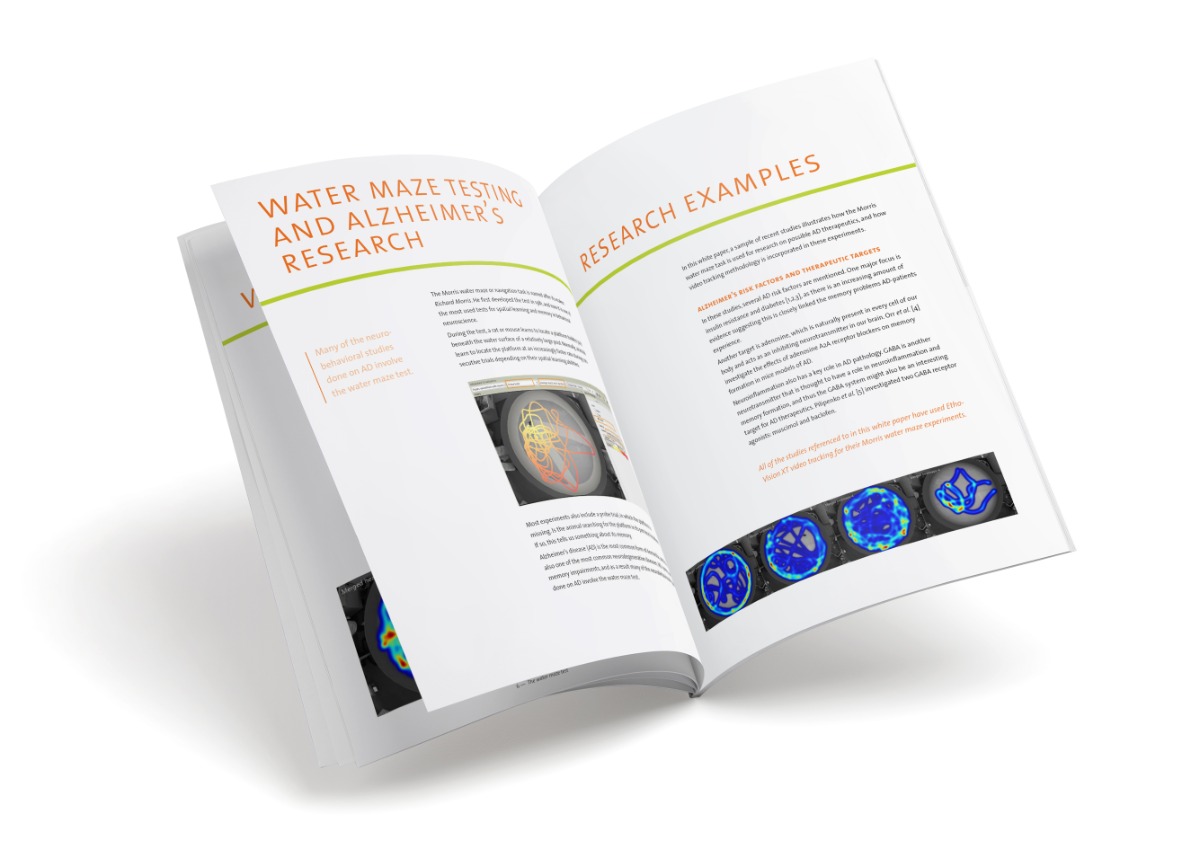
A standard protocol was followed: basic learning was testing using a hidden escape platform. Each mouse was given four swimming trials a day for five consecutive days. The mouse was released in the water and once it had found the platform is was allowed to stay there for fifteen seconds. If the mouse did not reach the platform in two minutes, it was guided to the platform. Escape latency, path length, and swimming speed were automatically measured with EthoVision XT. Exactly three days later a probe trial (no platform) was conducted for sixty seconds to measure memory retention. In this case, proportion of the time spent in the target quadrant (that previously held the platform) was measured with EthoVision XT.

Caffeine prevents memory impairment
All the mice in this experiment seemed to learn well. Their results improved over the five learning trials, and there were no differences in this improvement between the four treatment groups. The tau-pathology did not cause the ‘AD-mice’ to have any problems in learning. It did however result in specific memory issues. During the retention-trial, THY-Tau22 mice that did not receive any caffeine spent an equal amount of time in each of the quadrants. Interestingly, but as expected, the THY-Tau22 mice that did receive caffeine performed identically to wild type mice, spending more time in the target quadrant than any other. Therefore, caffeine did help maintain spatial navigation memory in tau-impaired mice. Caffeine did not seem to have any effect on the learning and memory of the wild-type mice.
Fewer tangles in the brain
Consistent with the results from the water maze retention trials, researchers found fewer tau-tangles in the brain cells of the THY-Tau22 mice that received caffeine compared to those who just got water. The level of caffeine taken by the mice in this experiment is considered to be equal to approximately two cups of coffee a day for humans. Chronic and moderate caffeine intake, at least in this study, did indeed seem to have a neuroprotective effect against this AD-related pathology,
So, there you have it, drink your coffee! Or tea, of course.
Reference
Laurent, C.; Eddarkaoui, S.; Derisbourg, M.; Leboucher, A.; Demeyer, D.; Carrier, S.; Schneider, M.; Hamdane, M.; Müller, C.E.; Buée, L.; Blum, D. (2014). Beneficial effects of caffeine in a transgenic model of Alzheimer’s disease-like tau pathology. Neurobiology of aging, article in press, doi: 10.1016/j.neurobiolaging.2014.03.027
Get the latest blog posts delivered to your inbox - every 15th of the month
more
Mice with Alzheimer’s disease walk well but remember poorly
A large number of genetically engineered mouse models are available to study different aspects of Alzheimer’s disease.
New method for early detection of Alzheimer's disease
The color x-maze can be used to pick up symptoms of Alzheimer's disease in AD-mice months before traditional cognitive tests.