Assessing gait in a mouse model for Leigh Syndrome
There is an unmet medical need to develop clinical effective treatments for Leigh Syndrome and other mitochondrial diseases. Today, Dr. de Haas shares some of her research insights.
Posted by
Published on
Thu 04 Apr. 2019
Topics
| CatWalk XT | Gait Analysis | Leigh Syndrome | Mitochondrial Diseases |

Today we have a guest blog post by Dr. Ria de Haas, on her preclinical research on Leigh Syndrome at the Radboud Center for Mitochondrial Medicine (Nijmegen, The Netherlands). Enjoy!
Leigh Syndrome
Mitochondrial diseases are heterogeneous multisystem disorders mostly caused by mutations affecting the oxidative phosphorylation (OXPHOS) system, which is involved in cellular energy production.
Leigh Syndrome is one of the OXPHOS deficiency disorders with an incidence of one in 77,000 live births and also known as sub-acute necrotizing encephalomyelopathy. Symptoms typically begin early in life, and include ataxia, loss of muscle strength, and neurodegeneration.
Abnormal gait in patients
Mitochondrial disease patients also show abnormal gait. The GAITrite is an electronic walkway used for patients and shows marked differences in gait between mitochondrial disease patients and controls 1,2. The observed gait differences correlate with markers of disease severity.
Treatment
Currently, there is an unmet medical need to develop clinical effective treatments for Leigh Syndrome and other mitochondrial diseases. Several potential new treatment strategies are currently under (pre-)clinical development; one of them is a novel small molecule KH176 3-6.
Mouse model for Leigh Syndrome
Because mitochondrial diseases are heterogeneous and multisystem disorders, it is important to investigate interactions at multiple levels (molecules, cells, organs and physiological functions). Therefore we are using the Ndufs4 knockout (Ndufs4-/-) mouse model which is a model for Leigh Syndrome and develops, similarly to the clinical situation, a fatal progressive neurodegenerative disease phenotype.
Symptoms include loss of motor function, ataxia, epilepsy, and short life span. This model is used to gain more insight into the pathogenesis of this disease and to evaluate the efficacy of novel therapeutics.

Methodology
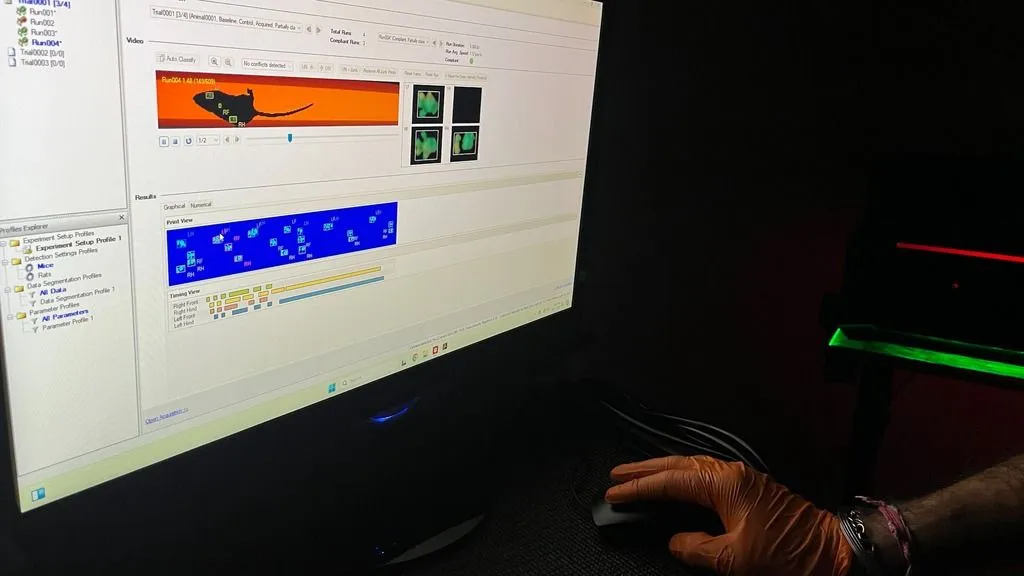
Previously we used more traditional readouts, such as rotarod and grip strength, to characterize this animal model. However, these readouts remain subjective and difficult to translate to the clinical situation. Therefore, in line with the GAITrite studies in patients, we used the CatWalk XT gait analysis to evaluate gait in the Ndufs4-/- mice 7.
We used the following protocol in our studies. Starting at the age of three weeks, mice were habituated to the CatWalk with their cage mates for 10 minutes during four consecutive days. By using this habituation protocol, the animals walked voluntary across the walkway without the use of any rewards. Weekly testing was performed individually, starting at four weeks of age. A session was successful after four compliant runs. A compliant run meets the run criteria set at a maximum run duration of 15s, with a maximum speed variation of 65%.
Abnormal gait in Ndufs4-/- mice
Marked genotype differences were noted between Ndufs4-/- and wild type mice in dynamic, static, coordination, and support parameters 7. Specifically walking speed variation, intensity of the individual limbs, number of patterns, and diagonal and lateral support were significantly affected in Ndufs4-/- compared to wild type mice.
RESOURCES: Read more about CatWalk XT
Find out how CatWalk XT is used in a wide range of studies and how it can elevate your research!
- Free white papers and case studies
- Customer success stories
- Featured blog posts
Treatment effect on gait
Therapeutic effects of KH176 on gait were evaluated in the Ndufs4-/- mice model 4. Long-term treatment with KH176 significantly reduced walking speed variation, increased intensity of the hind limbs, and number of patterns. Furthermore, diagonal and lateral support values were normalized.
This research indicates the ability of the CatWalk XT to sensitively and objectively assess gait abnormalities in Ndufs4-/- mice. CatWalk XT can be of great value in translational neuroscience, intervention studies, and drug development.
Learn more about gait analysis.
References
- Koene, S., et al., (2018). Quantification of gait in children with mitochondrial disease. J Inherit Metab Dis, 41(4), 731-740.
- Ramakers, R., et al., (2017). Quantification of gait in mitochondrial m.3243A > G patients: a validation study. Orphanet J Rare Dis,12(1), 91.
- Beyrath, J., et al., (2018). KH176 Safeguards Mitochondrial Diseased Cells from Redox Stress-Induced Cell Death by Interacting with the Thioredoxin System/Peroxiredoxin Enzyme Machinery. Sci Rep, 8(1), 6577.
- de Haas, R., et al., (2017). Therapeutic effects of the mitochondrial ROS-redox modulator KH176 in a mammalian model of Leigh Disease. Sci Rep, 7(1), 11733.
- Janssen, M.C.H., et al., (2019). The KHENERGY Study: Safety and Efficacy of KH176 in Mitochondrial m.3243A>G Spectrum Disorders. Clin Pharmacol Ther, 105(1), 101-111.
- Koene, S., et al., (2017). KH176 under development for rare mitochondrial disease: a first in man randomized controlled clinical trial in healthy male volunteers. Orphanet J Rare Dis, 12(1), 163.
- de Haas, R., F.G. Russel, and J.A. Smeitink. (2016). Gait analysis in a mouse model resembling Leigh disease. Behav Brain Res, 296, 191-198.
Images courtesy of author Dr. Ria de Haas, Radboud Center for Mitochondrial Medicine, Nijmegen, The Netherlands
Related Posts
How to use CatWalk XT and Incapacitance Tester in non-clinical pain research

True or false? 10 statements about gait research in rodents