What you need to know about the CatWalk gait analysis method
Let's take a little step back and talk about the history and technology of the CatWalk system. What was the reason of creating this gait analysis system for rats and mice? How does it work?
Posted by
Published on
Thu 29 Sep. 2022
Topics
| CatWalk XT | Gait | Gait Analysis | Locomotion | Mice | Parkinson's Disease | Rats | Spinal Cord Injury |

We have talked a lot about gait analysis lately, particularly about methods in animal research and the pros and cons of different systems. Today, I would like take a little step back and tell you about the history and technology of the CatWalk system.
CatWalk and models
Ok, so let’s start with the name. When hearing the word catwalk, you might think of fashion models strutting down a runway. Actually, that’s not that far off, as it is a runway made for rodents. Only we’re not talking fashion here, but scientific research.
In a nutshell: CatWalk XT is a system for automated gait analysis in rodents. It was originally created for research on rat models for spinal cord injury and is mostly used for rats and mice, but has expanded to other species, such as marmosets as well.

Turn-key gait analysis
The basic idea is that the animal freely traverses a walkway from one side to the other. This is a glass plate with green LEDs running through it. A technology called Illuminated Footprints captures the footprints made on the glass plate from a high-speed camera mounted underneath. (More about that later.)
This corridor is placed on a frame that also holds everything together, including a control unit to control the power. For convenience, this whole system is placed on lockable swivel casters.
Accompanying software analyzes the footprints recorded by the camera. A bunch of (read: more than 50) spatial and temporal parameters, not to mention some really cool 2D and 3D visuals, help you answer your research question.
CatWalk: the original
The original CatWalk was designed by Dr. Frank Hamers in 2001, because he and his colleagues wanted to find a better way to measure peripheral and central nervous system damage in spinal cord injury models. Like the current method, it was based on an animal traversing a glass walkway. Footprints were captured using white LEDs sent through the glass plate and an analog camera underneath.
Hamers and his colleagues collected data such as paw print area, swing duration, stride length, and base of support and compared these in two different models of spinal cord injury.
Using this new method, they were able to capture gait impairments introduced by two different rat models of spinal cord injury. No other typical method (like BBB scoring) came close, and so they quickly figured this way of gait assessment would be useful for other disease models as well.

Green light, red light
This is where we (Noldus) stepped in. By 2006 the system was ready for a commercial release, in 2009 it got a major upgrade. It now uses green light for the Illuminated Footprints technology. Green LED light is emitted in the glass walkway and footprints are illuminated where the paw touches the glass.
We also added a ceiling with red LED light to provide backlight for the video, creating a nice outline of the animal without disturbing it. And of course, the camera is now a high speed digital camera.
A system would not be a system without analyses, so the dedicated CatWalk XT software got some upgrades over the years, too. It automatically labels and classifies the prints, and has a lot of options for analysis and visualization. I won’t go into too much details here, because you can read all about on our website.

Extra motivation with a goal box
In 2011 we added the option of a goal box to provide some extra motivation for the animal to traverse the runway in a straight line. Because that’s the thing about free-run based systems: while it captures a more natural gait (instead of forced locomotion), the animal might linger or turn around on you.
Please enjoy this video I made when I just started to discover the joys of video editing. It's pretty old, but I think it gets the point across:
At Noldus, we want research to be efficient, in addition, we encourage appetitive reinforcement, so one of the ways to motivate the animal is by providing a goal. The reward could be the home cage, food, or (as Kristina Ängeby Möller shows in this white paper linked below) a social stimulus which often seems to work best, at least for rats.
RESOURCES: Read more about CatWalk XT
Find out how CatWalk XT is used in a wide range of studies and how it can elevate your research!
- Free white papers and case studies
- Customer success stories
- Featured blog posts
Another way to get a nice, straight, even run, is to adjust the side walls of the corridor. Adjust them to your animal so it has enough room to comfortably run, but is less likely to take a U-turn.
Visualize data
Automating your research makes things easy and objective, which is of course a major advantage of systems like CatWalk XT. What is sometimes overlooked is the presentation of all that data you can acquire.
I don’t know about you, but I am not one that can easily make sense of large tables with numbers. So, I like the visualizations in the CatWalk XT software a lot. It immediately gives a good sense of your data.
First, there’s the run video itself. You can replay it at different speeds and it will show you the classification of footprints as well.
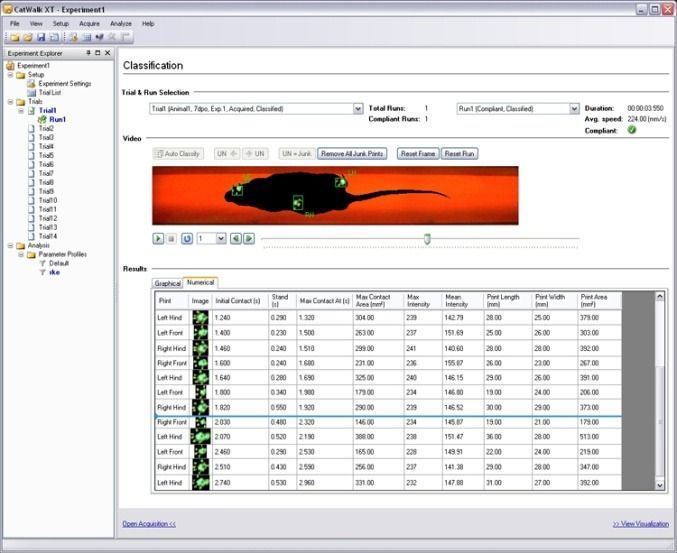
Then there are separate visualizations that show paw and run information in a two or three dimensional plane. What’s also really nice: you can export all in video files to use in lab meetings, presentations and defenses.
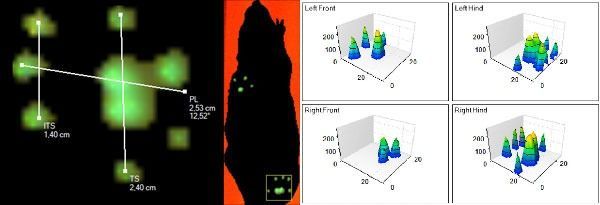
CatWalk XT provides a lot of data on the individual paws, the single runs, and overall trials. You pick what suits your research. This article would be way too long if I’d go into all of them, but trust me, there’s a lot.
What’s really nice: each parameter is described in full in the manual. In many cases, the exact formula is shown along with a publication to support it.
CatWalk XT in publications
Ok, so one last thing and then I promise to stop bragging about this system (for now). It has six times the number of publications than any other system. Six times! That amounts to over 2,400 publications and we are pretty proud of that.
Did I spark your interest? Then you might also like how our Product Manager Reinko Roelofs explains a bit more in this video.
References
Hamers, F.P.; Lankhorst, A.J.; Laar, T.J. van; Veldhuis, W.B.; Gispen, W.H. (2001). Automated quantitative gait analysis during overground locomotion in the rat: its application to spinal cord contusion and transection injuries. Journal of Neurotrauma 18(2), 187-201.
Pickett, K.A.; Schultz-Darken, N.; Bradfield, A.F.; Malicki, K.; Pape, B.; Ausderau, K.K.; Emborg, M.E. (2020). Spatiotemporal quantification of gait in common marmosets. Journal of Neuroscience Methods 330, 108517.
Related Posts

The many uses of the PhenoTyper
5 blogs on why you should use video tracking in behavioral research
