3 high impact papers about measuring anxiety in the Elevated Zero Maze
The elevated zero maze (EZM) is a variation on the elevated plus maze (EPM) and was created to eliminate the center region of the EPM. This blog highlights 3 studies that use the EZM to test for anxiety-like behavior
Posted by
Published on
Wed 06 Apr. 2022
Topics
| Anxiety | Avoidance Learning | Depression | Zero Maze |

image credit: Maze engineers
Measuring anxiety in the Elevated Zero Maze: 3 high impact papers
The elevated zero maze (EZM) is a variation on the elevated plus maze (EPM) and was created to eliminate the center region of the EPM. The EZM is an elevated ring-shaped arena with two open and two closed quadrants (with elevated walls), and has been described as valid test for anxiety-like behavior in rodents as early as 1994 [1].
Benefits of the elevated zero mazed compared to the elevated plus maze
The first benefit of the EZM, as already mentioned, is the removal of the center square. This takes away the availability of a logical starting point while also removing any ambiguity in the interpretation of the time spent in the center of the maze.
Secondly, Tucker and McGabe describe an experiment in which they compare the EPM and EZM in male and female C57BL/6J mice [2]. They found that the EZM encourages greater exploration of the anxiogenic regions (open quadrants), while also finding consistent outcomes over multiple sessions, while the EPM is advised to be only performed once per mouse to avoid learning and habituation to the maze, potentially masking anxiety-like behavior.
The elevated zero maze is gaining in popularity
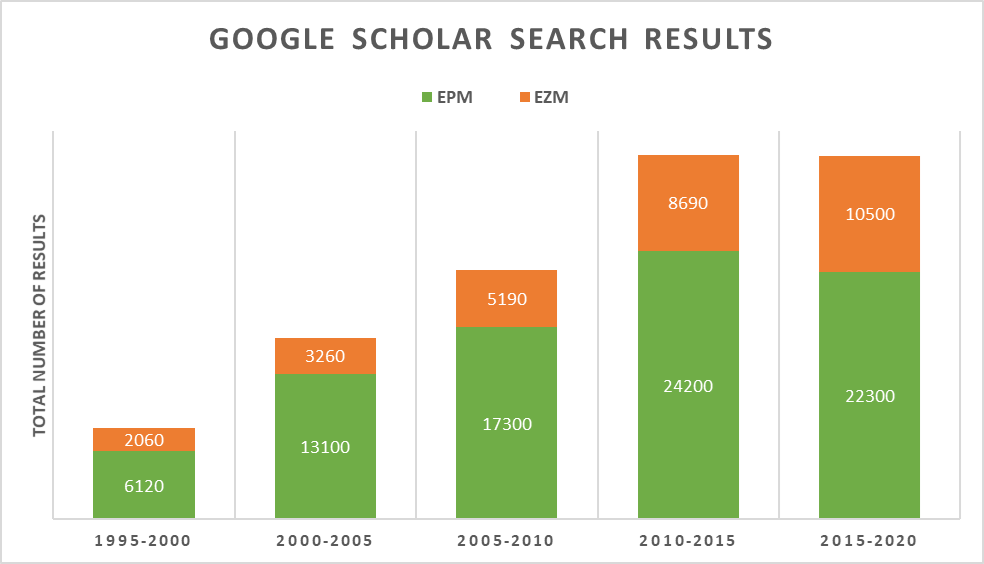
The EPM is still the most used behavioral test to assess anxiety-like behavior in rodents. The EZM has however been gaining somewhat in popularity over the last few decades, as seen by a graphic based on search result numbers in Google Scholar for the keywords: ‘Elevated plus maze’ and ‘Elevated zero maze’, summarized in 5-year periods.
Paper 1: Depression, not anxiety, is affected by the FTO gene.
Fat mass and obesity-associated protein, also known as alpha-ketoglutarate-dependent dioxygenase (FTO) was shown to be downregulated in the hippocampus of patients with Major depressive disorder (MDD) and in mouse models of depression. “Knockdown or knockout of Fto in the hippocampus induces depression-like behaviors, whereas overexpression of FTO has antidepressant effects” [3].
This study by Liu et al in ‘Nature communications’ showed that specifically depressive-like behaviors were affected by their FTO mouse model, while anxiety-like behaviors, as measured with the EZM, and growth/body weight changes were not observed. This is argued by the authors to be due to their FTO deficiency model presenting what is called: ‘nonanxious MDD’. This model actually creates a very nice opportunity to investigate the depressive symptoms of this model in a very isolated manner, meaning there is little influence by other behavioral confounders.
Paper 2: The alcohol deprivation effect (ADE) as a model of relapse-like drinking
Alcohol use disorder (AUD) is still a prevalent conditions nowadays, and has a very debilitating effect for individuals. Unable to regulate drinking behavior, increased feelings of cravings and chronic episodic relapsed towards heavy alcohol drinking make this an extremely difficult psychological and physiological disorder to treat. Stress, for example, has been identified as a major trigger for cravings and relapses.
The study by Chandler et al in ‘Neuropharmacology’ makes use of the Alcohol Deprivation Effect (ADE), a pre-clinical model for relapse drinking in rats [4]. They subject Sprague-Dawley rats to the ADE protocol, which introduces alcohol next to drinking water, but is taken away for a set period of time, whereafter it is re-introduced. This creates a relapse model, which can then be pharmacologically treated with different α5GABA-A receptor agonists. Anxiety was tested in the elevated zero maze using Noldus’ EthoVision XT video tracking.
Want to know more about tracking automated video tracking in EthoVision XT? We have a number of webinars that explain everything in great detail!
Paper 3: Identifying neural circuitry underlying avoidance behavior
Avoidance behavior is a fundamental part of survival in any animal species. External cues can trigger an avoidance (when that trigger indicates a potential risk), but also an approach (when there is a potential for a reward). These signals can be interpreted as conflicting, leading to an approach-avoidance conflict. The dorsomedial prefrontal cortex (dmPFC) has been linked to avoidance and decision-making under conflict. These are also key neural computations that seem to be altered in anxiety disorders.
The study by Loewke et al in ‘The Journal of Neuroscience’ uses fiber photometry recordings in both male and female mice during the elevated zero maze task [5]. They show heightened neural activity in frontostriatal but not frontoamygdalar projection neurons during exploration of the anxiogenic open arms of the elevated zero maze. This work identifies a specific corticostriatal circuit involved in encoding and controlling avoidance behavior, this will enable the development of more targeted symptom-specific treatments for anxiety disorders.
The future of the elevated zero maze
This blog highlights the gaining interest in the elevated zero maze, and its use in a number of interesting scientific papers. Its very simple, yet distinct, difference from the elevated plus maze is based on a valid argument; that the interpretation of an animals’ time spent on a center square can be somewhat ambiguous. It would be nice to see more studies that compare these two mazes, such as the paper by Braun et al [6].
Want to read more about the elevated plus maze? Check out this blog!
References- Shepherd, J. K.; Grewal, S. S.; Fletcher, A.; Bill, D. J.; Dourish, C. T. (1994). Behavioural and pharmacological characterisation of the elevated “zero-maze” as an animal model of anxiety. Psychopharmacology (Berl)., 116 (1), 56–64
- Tucker, L. B.; McCabe, J. T. (2017). Behavior of male and female C57Bl/6J mice is more consistent with repeated trials in the elevated zero maze than in the elevated plus maze. Front. Behav. Neurosci., 11, 13
- Liu, S. et al. (2021). Fat mass and obesity-associated protein regulates RNA methylation associated with depression-like behavior in mice. Nat. Commun. 2021 121, 12 (1), 1–13
- Chandler, C. M.; Reeves-Darby, J.; Jones, S. A.; Li, G.; Rahman, M. T.; Cook, J. M.; Platt, D. M. (2021). Modulation of relapse-like drinking in male Sprague-Dawley rats by ligands targeting the α5GABAA receptor. Neuropharmacology, 199, 108785
- Loewke, A. C.; Minerva, A. R.; Nelson, A. B.; Kreitzer, A. C.; Gunaydin, L. A. (2021). Frontostriatal Projections Regulate Innate Avoidance Behavior. J. Neurosci., 41 (25), 5487–5501
- Braun, A. A.; Skelton, M. R.; Vorhees, C. V.; Williams, M. T. (2011). Comparison of the elevated plus and elevated zero mazes in treated and untreated male Sprague-Dawley rats: Effects of anxiolytic and anxiogenic agents. Pharmacol. Biochem. Behav., 97 (3), 406
Related Posts
Our gut microbiota influences how we feel

Taking opioids during pregnancy: short- and long-term consequences in rats

