A new rat model for neonatal white matter injury
Preterm birth is a major problem in neonatal healthcare. Erik van Tilborg developed a new animal model to closely mimic this clinical situation, an important step in finding new treatment options.
Posted by
Published on
Tue 13 Feb. 2018
Topics
| EthoVision XT | Open Field | Rats | Social Behavior Research | The Observer XT | Video Observation | Video Tracking |
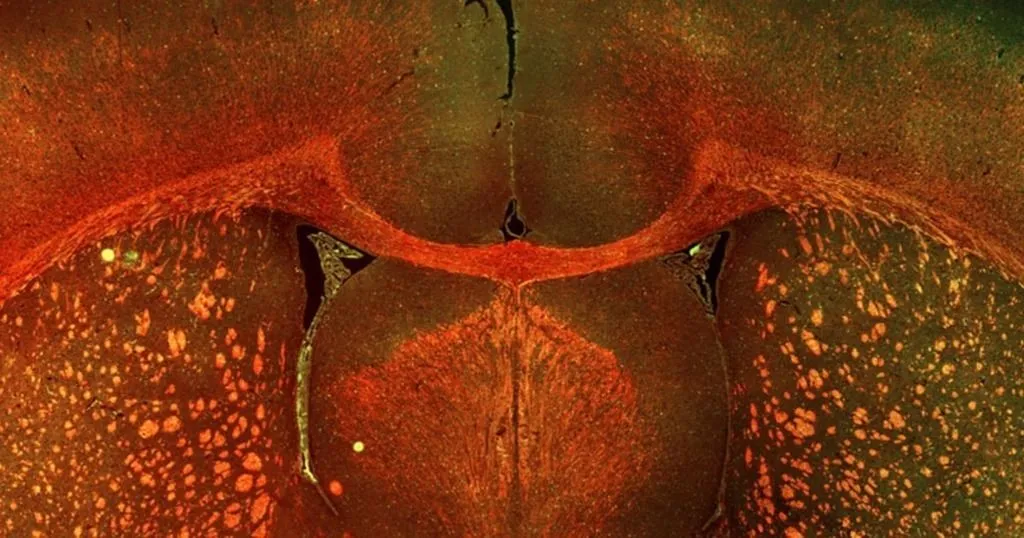
A guest blog post by Erik van Tilborg, PhD student in the research group of Cora Nijboer at the laboratory for Neuroimmunology and Developmental Origins of Disease (NIDOD), UMC Utrecht, The Netherlands.
White matter injury in preterm infants
Preterm birth is a major problem in neonatal healthcare. In the Netherlands 8% of all births occur prematurely, which means after a pregnancy of 37 weeks or less. Some babies are even born after a pregnancy of 24-27 weeks. These children are referred to as extremely preterm infants, and are often exposed to preterm birth-related health problems such as inflammation (due to an immature immune system and vulnerability to infections) and fluctuating oxygen levels (due to immature lungs, respiratory problems and underdeveloped vasculature in the brain).
In many preterm infants such complications, associated premature birth, cause diffuse injury to the white matter of the brain. Later in life, this type of white matter injury (WMI) leads to impaired cognitive performance, social dysfunctioning and decreased motor skills. Moreover, neonatal WMI is associated with an increased risk of psychiatric disorders, such as autism spectrum disorders, ADHD, schizophrenia and depression (reviewed in [1]).
Currently, no treatment options are available in neonatal clinics to prevent or cure WMI in preterm infants. Therefore, it is crucial that more research is performed to find out the cellular and molecular mechanisms that contribute to WMI in preterm infants, but also to find new treatment options.

A new rat model for neonatal WMI
In order to unravel the mechanisms underlying WMI and to develop new therapies, it is crucial to perform translational animal experiments. Mimicking neonatal WMI in rodents allows us to study this type of brain injury under controlled conditions and enables us to investigate new treatment options.
In order to develop a new rat model of neonatal WMI, we used to two insults that preterm infants are often exposed to and that are important risk factors to develop WMI: fetal inflammation and postnatal hypoxia. Careful analysis of the brains of rats exposed to these insults revealed that they show a pattern of brain injury that closely resembles the clinical situation of neonatal WMI [2,3]. To study the effects of the combination of these two insults on functional outcome, we used various behavioral paradigms.
Behavioral outcome in rats with neonatal WMI
To study motor performance, we used a rotarod task in which animals are placed on a beam that rotates at an increasing speed. In this task, it is measured how long it takes before the animal is not able to keep up anymore and falls down. This is considered to be a measure of motor performance. We observed that rats with WMI (induced by fetal inflammation and postnatal hypoxia) demonstrated a reduction in motor skills compared to control rats.
To study anxiety-like behavior, we studied the behavior of rats after being placed in an open field. During this task, the movement of the animals was tracked using Ethovision XT. It was measured how often the animals entered the middle ‘unsafe’ zone of the open field. Rats with WMI did this less often, which is considered a sign of anxiety as the animals spent more time in the ‘safe’ zone guarded by the walls of the field.
To determine the effects of combined fetal inflammation and postnatal hypoxia on social functioning, rats were placed in transparent cages in pairs and their social play behavior was scored using Observer XT. We observed that WMI animals showed a decrease in the initiation of social play interactions. Furthermore, using Observer XT the grooming behavior or the animals was studied. WMI rats showed an increase in the time spent on grooming – a sign of repetitiveness. Together, the observations of impaired social interactions and repetitive behavior are indicative of autism-like behavior.
From these behavioral experiments it can be concluded that WMI in rats is associated with impaired motor skills, anxiety-like behavior and autism-like behavior. This accurately reflects the clinical situation of WMI in preterm infants, who show similar behavioral problems later in life.
Future research
As the new animal model closely mimics the clinical situation of neonatal WMI, it may be used to study the cellular and molecular mechanisms that contribute to WMI in preterm infants and to develop novel therapies. Currently, we are using this animal model to test various treatment options by investigating whether they can reduce brain injury and improve functional outcome. Hopefully, this will lead to novel treatments that can improve outcome of vulnerable preterm infants for the rest of their lives. More detailed information regarding this study is available open access [3].
References
- van Tilborg E, Heijnen CJ, Benders MJ, van Bel F, Fleiss B, Gressens P, Nijboer CH (2016). Impaired oligodendrocyte maturation in preterm infants: Potential therapeutic targets.Progress in Neurobiology, 136, 28-49.
- van Tilborg E, van Kammen CM, de Theije CGM, van Meer MPA, Dijkhuizen RM, Nijboer CH (2017). A quantitative method for microstructural analysis of myelinated axons in the injured rodent brain.Scientific Reports, 7(1), 16492.
- van Tilborg E, Achterberg EJM, van Kammen CM, van der Toorn A, Groenendaal F, Dijkhuizen RM, Heijnen CJ, Vanderschuren LJMJ, Benders MNJL, Nijboer CHA. Combined fetal inflammation and postnatal hypoxia causes myelin deficits and autism-like behavior in a rat model of diffuse white matter injury.Glia, 66(1), 78-93.
Related Posts

Primate behavior - cracking the nut, fur rubbing & mating behavior

Bed bug behavior - What smell can tell

