What is the connection between depression and rheumatoid arthritis?
Is depression in patients with rheumatoid arthritis caused only by the pain, or is something else also going on?
Posted by
Published on
Thu 16 Feb. 2023
Topics
| Arthritis | Depression | EthoVision XT |
Rheumatoid arthritis (RA) is an autoimmune disease in which the joints swell up, causing pain and damage. An autoimmune disease is one where your own immune system attacks your own body, somehow confusing it with something like a disease-causing bacteria.
There are about 100 autoimmune diseases including diabetes type I, Crohn's disease and multiple sclerosis (MS). Whereas rheumatoid arthritis is an autoimmune disease, osteoarthritis is age-related. There are also many other less common forms of arthritis.
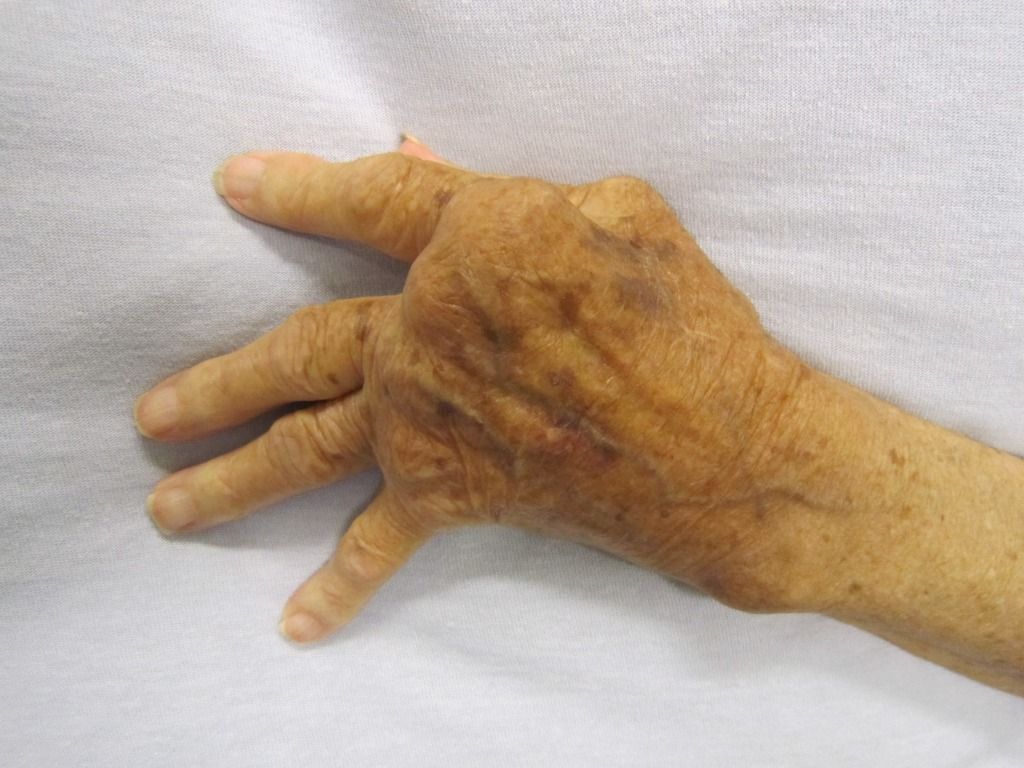
Hand with rheumatoid arthritis (© James Heilman).
Depression in rheumatoid arthritis patients
Many RA patients also experience depression, about two to four times as many as in the general population [2]. The large variance is due to the different ways of measuring if someone has depression.
At first sight, that is hardly surprising. If you are in pain all the time, then that is likely to cause a negative mood and that can cause depression. Rheumatoid arthritis patients can also experience handicaps due to not being able to use their joints properly, which will also make depression more likely.
However, there are some clues that there is more to it than that. For instance, antibiotics can sometimes relieve the symptoms of RA. That is strange; antibiotics kill bacteria, but RA is not caused by bacteria. How is that possible?
Gut bacteria
Although we tend to think of bacteria as being bad things, causing diseases, actually we would not be alive without healthy bacteria in our digestive tract (and elsewhere). Without those bacteria, we could not take up some of the nutrients that we need.
When we take a course of antibiotics, the bacteria in our gut are also killed, although the gut flora quickly re-establishes itself afterwards. Perhaps the effect of antibiotics on RA and the prevalence of depression in RA patients could have a common cause?
Gut-brain-microbiota axis
That common cause could be found in the gut-brain-microbiota axis. For many years, it has been known that there is an important interaction between the gut and brain for maintaining homeostasis. Homeostasis means regulating that the body so that it maintains a constant state with respect to its internal physical and chemical conditions.
This role of the gut is so important that its nervous system (called the enteric nervous system) has been referred to as the "second brain", also because it can act independently of the body's other nervous systems (parasympathetic and sympathetic) which are controlled by the brain.
More recently, it has become apparent that the microbiological population of the gut (that is, the bacteria and other microorganisms) also play a crucial role in that relationship, so that it is possible to speak of a microbiota-gut-brain axis.
The microbiota and the brain communicate with each other using various routes including the immune system, the vagus nerve and the nervous system of the gut. Various metabolites produced by the microbiota have an effect on those organs, which then have an indirect effect on the brain.
T cells
For example, lymphoid tissue associated with the gut (called Peyer's patches) can receive signal molecules from gut bacteria and those cells then determine the balance between T-helper and T-regulatory cells. T cells are a sort of lymphocyte (white blood cell), which are a key component of the immune system.
When T-helper cells are stimulated by an antigen (e.g. a pathogenic bacteria) they release chemical messengers (called cytokines) which stimulate other lymphocytes to attack antigens. T-regulatory cells control the immune reactions. So if the numbers of T-regulatory and T-helper cells get out of balance, then the whole system will not work correctly.
It is known that the gut-brain-microbiota axis can play an important role in depression [3]. T-regulatory cells are reduced in patients with major depressive disorder. That shows the possibility of a direct connection between gut bacteria and depression.
Seeing RA is an autoimmune disease, possibly the depression frequently experienced by RA patients could be not just due to the pain, but might be a direct effect caused by the gut-brain-microbiota axis.
Transplant experiment
Yaoyu Pu and colleagues [1] wanted to investigate if that might be a real connection. Could certain gut bacteria be disrupting the immune system and being an additional cause of why more RA patients often have depression than the general population?
That is quite hard to answer, because it is one thing to show a correlation between RA and depression, but that doesn't mean that one causes the other. The cause and effect could be the other way around, e.g. a faulty immune system affecting the gut bacteria and the depression being purely caused by pain. They tackled this problem with an ingenious approach, combining laboratory mice and people suffering from RA.
Fecal transplants
They transplanted feces from human patients with RA and from a healthy control group, into laboratory mice. Then they measured if there were any differences between the behaviors of the two groups. If the feces from RA patients caused the mice to show depressive-like behaviors, then this proves that it is a real causal effect, not just a correlation.
They measured a number of different behaviors in the mice using EthoVision XT. EthoVision is an advanced video tracking system which can accurately measure movements and a range of behaviors in experimental animals.
Results: testing depression in mice
Locomotion in an open field
Firstly, they looked at locomotion. That is simply how much the mice moved around in an enclosed area with nothing in it for ten minutes. They found no difference between the two groups. Lack of activity can be a symptom of depression, but it is also affected by all sorts of other factors like the health status of an animal.
Learned helplessness with the tail suspension test
Secondly, they carried out two tests related to a concept called learned helplessness. Normally if you try something, and you don't succeed, and then you are faced with the same problem again, you will try again. However, if you are suffering from depression, you are more likely to give up sooner, when you are faced with a problem that you have previously failed to solve.
In the tail suspension test, a mouse is suspended by its tail. This does not hurt it, but it does make it anxious. The mouse will try to escape, but after a while, it will give up and become immobile. The idea is that becoming immobile earlier is a measure of depression. Yaoyu Pu and colleagues found that the mice that had received fecal transplants from RA patients were immobile for longer than the mice with transplants from healthy controls.
The forced swim test
The other test related to depression is the Porsolt forced swimming test. In this test, mice are placed in a glass cylinder, half filled with water. At first, the mice try to escape, but after a while, when they fail to climb the cylinder's walls, they give up. The mice which had received the fecal transplants from RA patients gave up significantly earlier than the control group.
Sucrose preference test
The final test that they did was the sucrose preference test. In that test, the two mice groups are fed water & 1% sucrose (sucrose solution) for 48 hours, which was followed by four hours with neither water nor food. After that, they had acces to a sucrose solution for one hour. The amount of sucrose solution consumed in that hour is measured.
The idea is that the more pleasure they get from the sweet sucrose, the more they will drink. A symptom of depression is a reduced ability to experience pleasure, in this case reduced sucrose preference, also called anhedonia. In the RA treatment group, sucrose preference was significantly lower compared to the control group.
Gut microbiota
Both groups of mice were treated with antibiotics before the experiment, so their gut flora would be severely reduced, such that they can be assumed to have a similar starting point. After the fecal transplant, the mice receiving the feces from RA patients had a completely different gut flora (bacterial composition) than the mice receiving feces from the control group. The RA group also had a much greater diversity of microbiota than the controls.
Conclusions
The conclusion of Yaoyu Pu his colleagues was that the data indicate that fecal transplants from rheumatoid arthritis patients could activate abnormal differentiation of T cells by changing the gut microbiota in the antibiotic treated mice.
The T-regulatory cells protect against depressive behaviors in rodents. This study showed that alterations in the gut microbiota can cause a decrease in T-regulatory cells, leading to systematic inflamation, which in turn can lead to depressive like behaviors.
Their final conclusion was the traditional one, that more research is needed. The study indeed raises all sorts of interesting questions. Mice and human gut anatomy are very different [4]; how much did that affect the results? Laboratory mice are kept in a sterile environment and fed a very uniform diet, might that cause the gut flora to develop in a different way?
What happens if you compare fecal transplants from people with and without depression (but with RA)? How much of the depression is caused by the pain of RA and how much by the gut bacteria?
Yaoyu Pu's paper is actually more complex than the simplified version presented here, with additional experiments and measurements. It is available open access and I encourage you to take a look [1].
References
1. Yaoyu Pu, Qiuping Zhang, Zhigang Tang, Chenyang Lu, Liang Wu, Yutong Zhong, Yuehong Chen, Kenji Hashimoto, Yubin Luo1and Yi Liu (2022). Fecal microbiota transplantation from patients with rheumatoid arthritis causes depression-like behaviors in mice through abnormal T cells activation. Translational Psychiatry 12:223. https://doi.org/10.1038/s41398-022-01993-z
2. Margaretten M, Julian L, Katz P, Yelin E. Depression in patients with rheumatoid arthritis: description, causes and mechanisms. Int J Clin Rheumtol. 2011;6:617–23.
3. Cryan JF, O’Riordan KJ, Cowan CSM, Sandhu KV, Bastiaanssen TFS, Boehme M, et al. The microbiota–gut–brain axis. Physiol Rev. 2019;99:1877–2013. DOI: 10.1152/physrev.00018.2018
Related Posts

Anxiety is ancient – how crayfish resemble vertebrates
Zinc deficiency, depression and electrical signals in the brain

