Furthering behavioral studies for understanding microbiota-brain insights
How and to what extend do microbes in the gut affect the brain and behavior? Discover how they do this kind of research at UCLA.
Posted by
Published on
Thu 19 Aug. 2021
Topics
| Automating Behavioral Observations | Behavioral Research | EthoVision XT |
Today we are pleased to share with you a guest blog post by Dr. Christine Ann Olson from University of California, Los Angeles (UCLA). Enjoy reading!
Behavioral Implications in Microbiome-Brain Research
The gut microbiota is collectively defined as the microbes present in the gastrointestinal tract. Current research demonstrates in rodent models that disrupting the microbiota through germ-free rearing or antibiotic depletion results in changes to cognitive, social, communicative, and stress-related behaviors (Vuong et al., 2017). While these models are of course artificial, these findings raise the question: to what extent to microbes in the gut affect the brain and behavior?
Since many neurologic disorders, including neurodevelopmental and neurodegenerative disorders, have unknown or multifaceted origins, one question is whether the gut microbiota could be partially responsible for disease states. Our laboratory (Elaine Hsiao, PhD) at UCLA focuses on whether microbes affect these disease states in preclinical models, and if so, how microbes exert noted effects. An ever-present question in this nascent area of research is how to uncover mechanisms for noted effects, which circuits are involved, and why there are differences across papers in response.
The Importance of Studying Behavior...with a Keen Eye
Studying animal behavior means evaluating an integrated response to a host of internal and external stimuli and involves an integration of sensation, response, motor function, learning and memory, and novelty versus habituation. Given the integrative and emergent nature of behavior, it is a useful tool to understand how the brain responds to experimental manipulation.
In a recent opinion piece, Lewis and authors argue that while quantitative behavioral metrics have their rightful and important place in studying the brain, both qualitative analysis and standardized procedures for understanding and contextualizing behavioral research are essential to parsing circuit-level control of behavior (Lewis et al.; Lewis et al., 2021). We will be evaluating this opinion in the context of microbiota-brain interactions.
Leveraging Quantitative and Qualitative Metrics to Understand Microbiome-Brain Research
As referenced before, the behavioral findings from the consequences of microbiota depletion are mixed (Vuong et al., 2017). For example, for mice raised germ-free, in tasks measuring sociability, sometimes germ-free mice exhibited increased sociability compared to specific-pathogen free (SPF) mice, and sometimes, germ-free rodents exhibited reduced sociability compared with SPF rodents.
While it is possible age is a factor in this case because various ages were tested across these studies, the animals tested were adults, and one may think that the age should not be a significant factor within certain bounds (i.e. the most significant disruption in this case is the germ-free rodents not being raised with a microbiota). Other tests are more consistent across behavioral testing paradigms, such as when germ-free rodents are compared with SPF mice for locomotion and stress-related behaviors: across a variety of tasks, germ-free rodents exhibit increased exploratory behavior compared with SPF rodents. Does this consistency reflect a more consistent circuit which is targeted, or something more complicated?
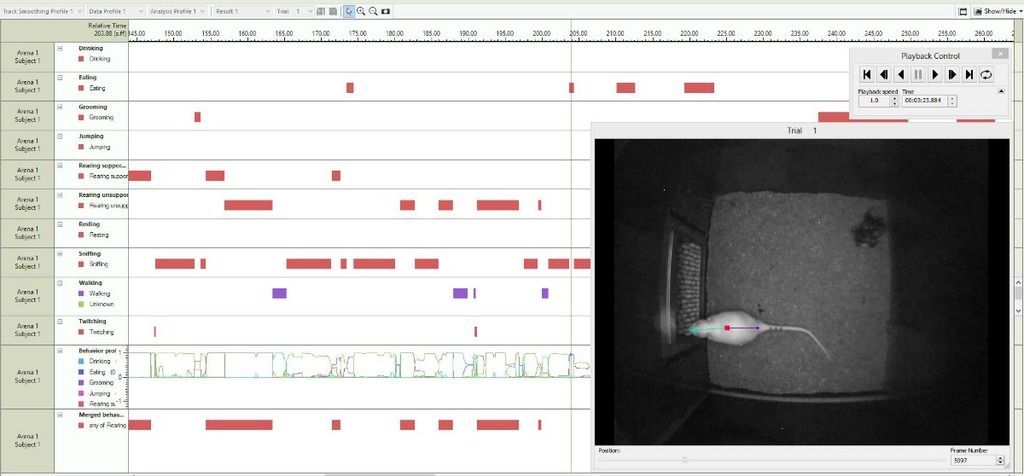
EthoVision XT - automated behavior recognition
Improving consistency and reliability of data
Taking the advice from Lewis et al., we should proceed with humility and test a variety of circuits in the future to pinpoint why this task yields consistent results. Additionally, using consistent and automatic behavioral tracking methods such as the suite of tools in EthoVision XT, including automated behavior tracking for tasks like grooming, rearing, drinking, sniffing, reliable velocity and distance metrics, and custom behavior modules can help with consistency and reliability of measuring results.
It is possible that conflicting results more broadly in behavioral neuroscience and more specifically in the context of microbiota-brain interactions can be explained by a lack of standardized interpretations of behavioral experiments and by a lack of reliance on quantitative and qualitative behavior together to draw an informed conclusion.
References
Lewis, A.S., Calipari, E.S., and Siciliano, C.A. (2021). Toward Standardized Guidelines for Investigating Neural Circuit Control of Behavior in Animal Research. eNeuro 8.
Vuong, H.E., Yano, J.M., Fung, T.C., and Hsiao, E.Y. (2017). The Microbiome and Host Behavior. Annual review of neuroscience 40, 21-49.
Related Posts

Ladybugs and lacewings do not spy on their prey’s alarm pheromone
A new rat model for neonatal white matter injury

