Understanding face perception in ASD
Researchers at KU Leuven want to gain more insight into whether or not there are differences in the implicit abilities of children with ASD to detect faces, different identities, and different expressions.
Posted by
Published on
Mon 06 Nov. 2017

This blog post is a guest post by Stephanie Van der Donck from the Center for Developmental Psychiatry of KU Leuven in Belgium. The study Stephanie writes about, investigates different processing abilities in children with and without autism spectrum disorder (ASD). She explains why they choose to apply an innovative scalp electroencephalography (EEG) approach combined with fast periodic visual stimulation (FPVS).
Processing faces
The human face is a highly familiar, complex, and multidimensional visual pattern that provides a wealth of information about someone (e.g. his or her identity, gender, age, emotional state, etc.). Recognizing people and accurately interpreting non-verbal cues like facial expressions are required to successfully interact with others.
Individuals with autism spectrum disorder (ASD) are characterized by deficits in social communication and interaction (American Psychiatric Association, 2013). Many individuals with ASD show impairments in processing faces (e.g. Webb, Neuhaus, & Faja, 2016; Weigelt, Koldewyn, & Kanwisher, 2012). They show impaired sensitivity to social communication cues conveyed as, among others, difficulties with non-verbal communicative behaviors and deficits in expressing and recognizing facial affect (Constantino & Charman, 2015).
Face processing abilities in people with ASD have already been widely studied, both implicitly (e.g. brain imaging studies) and explicitly. However, results are mixed and inconsistent. A partial explanation for this variability could be the use of explicit behavioral tasks. This is because performances of these tasks can be influenced by several factors beyond face processing, such as attention and motivation. Implicit measures do generate information that is more consistent across studies.
However, they usually yield weak effects and low signal-to-noise ratio, which requires long recordings, complex measures, and/or time-consuming analysis. This large variability and inconsistency in results impedes defining and understanding the nature of face processing impairment, especially at the individual level.
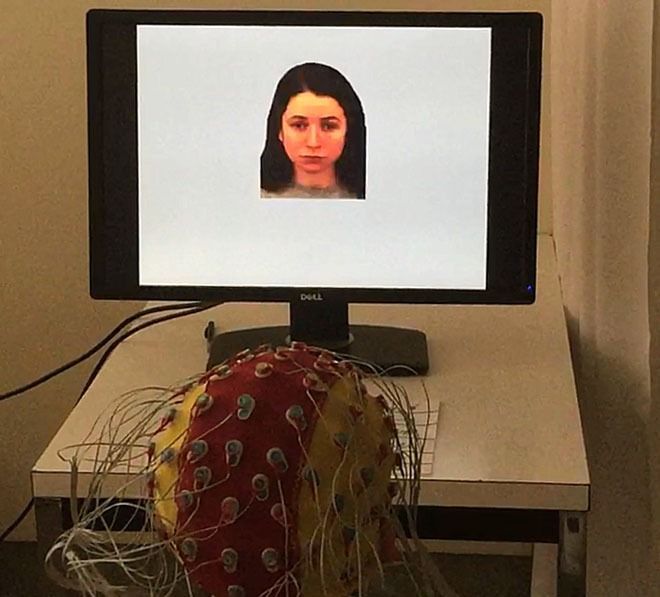
Fast periodic visual stimulation
To address the limitations of the methodologies used in previous studies, we apply an innovative scalp electroencephalography (EEG) approach combined with fast periodic visual stimulation (FPVS). With FPVS, a flickering stimulus is generated by rapidly presenting images (e.g. at 6 Hz). The general principle of FPVS is that it elicits a brain response (i.e. a steady-state visual evoked potential) at exactly the same frequency as the frequency of visual stimulation (for review, see Norcia et al., 2015).
We use FPVS in an oddball paradigm where we periodically enter oddball images every 5th image (i.e. 6 Hz/5 = 1.2 Hz). The quick transition from the base images to the oddball images measures a discrimination response by forcing the participants to detect rapid changes in, for example, identity and/or expression.
We also added an orthogonal task: the participants have to press a button as quickly as possible when they detect a change of color in the fixation cross that is presented on the screen. This orthogonal task ensures that all neural responses will be evoked implicitly.
FPVS EEG has several advantages compared to approaches used in previous studies. By adding the orthogonal task, FPVS EEG yields implicit measures. Furthermore, these measures are objective, since the responses can be identified at a predefined frequency ( the oddball frequency and its harmonics).
Consequently, getting a response at that predefined frequency and its harmonics allows direct quantification of the response of interest. Data can be collected within a few minutes (e.g. 4 sequences of 40 seconds) and can be analyzed without any complex post hoc comparisons of responses evoked by standard and deviant stimuli as in standard ERP studies.
Implicit vs explicit sensitivity to social cues
In this stage of our study, we will investigate different processing abilities in children with and without ASD. By using FPVS, we want to gain more insight into whether or not there are differences in the implicit abilities of children with ASD to detect faces, different identities, and different expressions in comparison to typically developing matched controls.
Besides these implicit measures, we also added several explicit identity and expression recognition tasks. The explicit tasks are implemented in combination with FaceReader software and eye tracking. This way, we want to assess if the strategies used to scan faces and the level of facial mimicry could be linked to potential differences between children with and without autism in detecting and/or recognizing faces and expressions.
Through the combination of implicit and explicit measures, we hope to get a better idea of the extent to which children with and without autism are able to detect and process identities and expressions.
References
- American Psychiatric Association. (2013). Diagnostic and statistical manual of mental disorders (5th ed.) Arlington, VA: American Psychiatric Association
- Constantino, J.N., & Charman, T. (2015). Diagnosis of autism spectrum disorder: reconciling the syndrome, its diverse origins, and variation in expression. The Lancet Neurology, 15(3), p.279-291. doi: 10.1016/S1474-4422(15)00151-9
- Norcia, A.M., Appelbaum, L.G., Ales, J.M., Cottereau, B.R., & Rossion, B. (2015). The steady-state visual evoked potential in vision research: A review. Journal of Vision, 15(4).
- Webb, S.J., Neuhaus, E., & Faja, S. (2016). Face perception and learning in autism spectrum disorders. The Quarterly Journal of Experimental Psychology. doi: 10.1080/17470218.2016.1151059
- Weigelt, S., Koldewyn, K., & Kanwisher, N. (2012). Face identity recognition in autism spectrum disorders: a review of behavioral studies. Neuroscience and Biobehavioral Reviews, 36, p. 1060-1084. doi: 10.1016/j.neurobiorev.2011.12.008
Contact details
Stephanie Van der Donck, Center for Developmental Psychiatry, Katholieke Universiteit Leuven, Belgium ([email protected])
Prof. Dr. Bart Boets, Center for Developmental Psychiatry, Katholieke Universiteit Leuven, Belgium ([email protected])
Related Posts

Become and stay aware about children with autism

Emotion analysis can be beneficial to researchers in decision making

