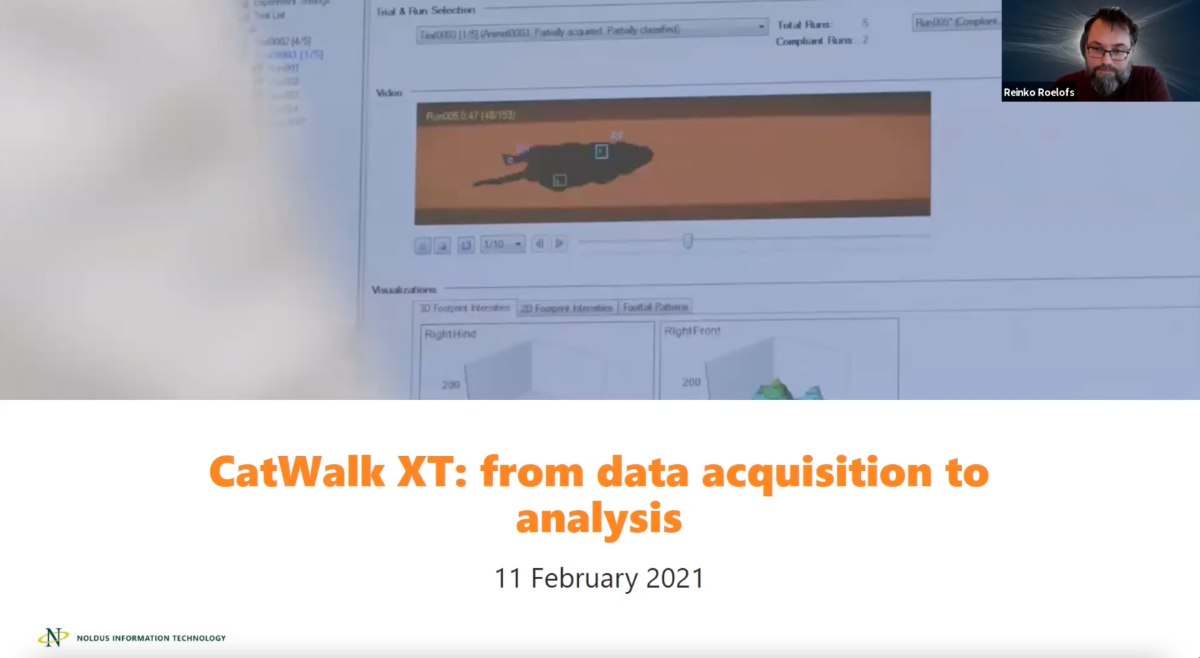
CatWalk XT
Resources
Webinar
If you are interested in knowing more about different models of peripheral nerve injury that can be studied with CatWalk XT and essential prerequisites for adequate data acquisition, you can take a look at a recording of a free webinar, where Dr. Johannes Heinzel explains all about it.
You can also request for more information or a free demo to get to know CatWalk XT system better and discuss with our scientific consultant about your research plans and wishes.
Recent videos
Neurodegenerative disease studies
Prof. Diana Price, NeuroPore Therapies, San Diego
Goal box CatWalk XT
CatWalk XT includes a goal box to motivate your animal to cross the walkway in a consistent manner
CatWalk XT
Gait analysis
Installation of CatWalk XT
Preview only
Automatic Footprint Classification
It's extremely fast!
Download our white papers
Phase Dispersions
Insights on the inter-limb coordination can provide you with valuable and important data for your research. Do you know that CatWalk XT calculates this data automatically? In this white paper we will take a closer look at Phase Dispersions values and how they are calculated, using simple and practical examples.
CatWalk XT and TBI mice
The CatWalk XT gait analysis system can be an incredibly useful tool in assessing the subtle motor deficits exhibited by mice post-TBI, making it ideal for detecting deficits that might not be readily observable to researchers. Read tips about how to use CatWalk XT for TBI models in this free white paper.
Getting the perfect runs
Familiarizing your rats with the CatWalk XT system before you start your experiment makes your job a lot easier. So how do you familiarize and motivate your rats? Kristina Ängeby Möller has a lot of experience with pain research and gait analysis in rats, and shares some tips and tricks in this free white paper!
Download our case studies
References
Here is a selection of recent papers that mention the use of CatWalk XT. There are many more, and if you are wondering if CatWalk XT has been used for a specific disease model, feel free to contact us or click at all publications below. If you feel your paper should be on this list, please let us know at [email protected]!
Catwalk Parameters
- Timotius, I.K.; Roelofs, R.F.; Richmond-Hacham, B.; Noldus, L.P.; von Hörsten, S.; Bikovski, L. CatWalk XT Gait Parameters: A Review of Reported Parameters in Pre-Clinical Studies of Multiple Central Nervous System and Peripheral Nervous System Disease Models. Front. Behav. Neurosci. 2023, 17, 1147784.
Spinal Cord Injury
- Zheng, G.; Zhang, H.; Tail, M.; Wang, H.; Walter, J.; Skutella, T.; Unterberg, A.; Zweckberger, K.; Younsi, A. Assessment of Hindlimb Motor Recovery after Severe Thoracic Spinal Cord Injury in Rats: Classification of CatWalk XT® Gait Analysis Parameters. Neural Regen. Res. 2023, 18, 1084.
Nerve Injury
- Dolińska, A.; Górka, D.; Trzęsicki, M.; Białoń, N.; Górka, M. The Role of Training and Hyperbaria in the Rehabilitation Process after Crushing Injury of the Sciatic Nerve in Mice - Assessment of Functional Parameters Using the CATWALK XT Platform. J. Educ. Health Sport 2023, 13, 150–159, doi:10.12775/JEHS.2023.13.04.016.
Arthritis
- Ko, J.-Y.; Wang, F.-S.; Lian, W.-S.; Fang, H.-C.; Kuo, S.-J. Cartilage-Specific Knockout of MiRNA-128a Expression Normalizes the Expression of Circadian Clock Genes (CCGs) and Mitigates the Severity of Osteoarthritis. Biomed. J. 2023, 100629.
Alzheimer's disease
- Brody, A.H.; Nies, S.H.; Guan, F.; Smith, L.M.; Mukherjee, B.; Salazar, S.A.; Lee, S.; Lam, T.K.T.; Strittmatter, S.M. Alzheimer Risk Gene Product Pyk2 Suppresses Tau Phosphorylation and Phenotypic Effects of Tauopathy. Mol. Neurodegener. 2022, 17, 32, doi:10.1186/s13024-022-00526-y.
Ataxia
- Haas, E.; Incebacak, R.D.; Hentrich, T.; Huridou, C.; Schmidt, T.; Casadei, N.; Maringer, Y.; Bahl, C.; Zimmermann, F.; Mills, J.D.; et al. A Novel SCA3 Knock-in Mouse Model Mimics the Human SCA3 Disease Phenotype Including Neuropathological, Behavioral, and Transcriptional Abnormalities Especially in Oligodendrocytes. Mol. Neurobiol. 2022, 59, 495–522, doi:10.1007/s12035-021-02610-8.
Brain Injury
- Walter, J.; Mende, J.; Hutagalung, S.; Grutza, M.; Younsi, A.; Zheng, G.; Unterberg, A.W.; Zweckberger, K. Focal Lesion Size Poorly Correlates with Motor Function after Experimental Traumatic Brain Injury in Mice. PLOS ONE 2022, 17, e0265448, doi:10.1371/journal.pone.0265448.
Parkinson's disease
- Zhu, Q.; Song, J.; Chen, J.-Y.; Yuan, Z.; Liu, L.; Xie, L.-M.; Liao, Q.; Ye, R.D.; Chen, X.; Yan, Y.; et al. Corynoxine B Targets at HMGB1/2 to Enhance Autophagy for α-Synuclein Clearance in Fly and Rodent Models of Parkinson’s Disease. Acta Pharm. Sin. B 2023, 13, 2701–2714, doi:10.1016/j.apsb.2023.03.011.
Stroke
- Pawletko, K.; Jędrzejowska-Szypulka, H.; Bogus, K.; Pascale, A.; Fahmideh, F.; Marchesi, N.; Grajoszek, A.; Gendosz de Carrillo, D.; Barski, J.J. After Ischemic Stroke, Minocycline Promotes a Protective Response in Neurons via the RNA-Binding Protein HuR, with a Positive Impact on Motor Performance. Int. J. Mol. Sci. 2023, 24, 9446.
Pain research
- Yerofeyeva, A.-M.V.; Pinchuk, S.V.; Rjabceva, S.N.; Molchanova, A.Y. The Role of Cannabinoid CB1 Receptors in the Antinociceptive and Reparative Actions of Mesenchymal Stem Cells in Rats with Peripheral Neuropathic Pain. Ibrain, doi:10.1002/ibra.12129.
Download our product overviews
Recent blog posts

How to characterize behavioral phenotypes in a behavioral analysis facility
Introducing the Behavioral Analysis Facility. Researchers evaluate the behavioral and functional activities of new pharmacological drugs using diverse functional tests. Learn more about their recent projects.
How to let rats run perfectly
Letting animals walk freely in gait research. At Noldus, we strongly believe this is the way to go, so that is how our CatWalk XT system works.
 English
English German
German French
French Italian
Italian Spanish
Spanish Chinese
Chinese