Track3D
Using Track3D for fish
Video tracking has significant advantages over manual recording of behaviors; think of accurate information about swimming speed, mobility, angular movement, and duration the animal was within a zone. It also makes it possible to perform uninterrupted observations over longer periods of time.
3D over 2D
While ‘normal’ tracking in 2D offers this advantages in studies on all kinds of animals, some animals fly or swim, implicating that there can be circumstances in which 3D tracking is very desirable.
For instance, in the novel tank test, fish may exhibit the behavior of thigmotaxis (swimming close to the walls) or diving. Tracking in three dimensions is very useful to distinguish the two behaviors. Likewise, although measurements of velocity and distance moved carried out in 2D will often give a good approximation, there are occasions in which information can be lost, simply because a fish is swimming straight towards the camera while no movement is recorded.
Studies have indeed indicated that the added accuracy of 3D tracking as opposed to 2D tracking reveals differences between treatment groups that would otherwise be left undiscovered (zebrafish studies done at the Kalueff Lab - see the references below or this blog post.
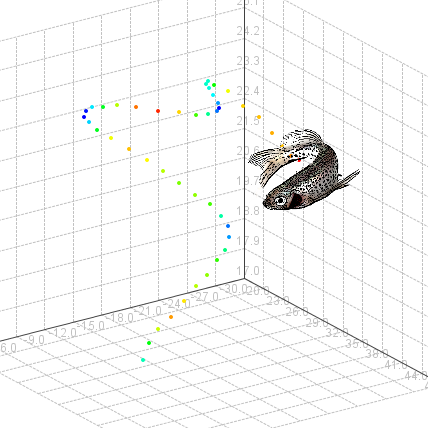
An experiment in Track3D
A typical Track3D experiment starts with video tracking the fish from two camera views using EthoVision XT. Track3D processes this data to create 3D visualizations and statistics of the swimming pattern of fish.
Read more about how Track3D works here.
Essential calibration
For an accurate calculation of the 3D trajectory of a fish from the tracking data of two video images, it is essential to calibrate. Our Track3D system includes a calibration frame that is easy to use by simply placing it in the water tank. This calibration step is essential because you are dealing with perspective and light refraction of the water. These two factors need to be taken into account, otherwise your data will be inaccurate. Calibration will make sure that the data from the two video cameras is accurately matched up to reconstruct the animal’s three-dimensional swimming path reliably.
Please note that the calibration frame is designed based on your research needs. You can use this frame with different tanks if their sizes are approximately the same. As long as your set-up is fixed and you are not moving your cameras, you do not need to recalibrate between trials or experiments.
In this video you can see how calibration with Track3D works.
Track3D is highly accurate
In contrast to most other 3D systems, the 3D reconstruction of the animals pathway is very accurate with Track3D. To be precise: with zebrafish test tanks, calibration error is generally around 0.5 mm. This is a good start for accurate 3D positioning of the subject. Uncertainty of 3D positioning of zebrafish is around 2-3 pixel in an image of 640x480 pixel. For details, see references below (Stewart et al., 2015).
This video shows how Track3D visualizes and analyzes the circling behavior of zebrafish.
Examples of research results
Example 1 – Visualization of the 3D trajectories. Compared with controls, treatment with a glutamatergic antagonist like PCP evokes a complex trajectory in zebrafish, with rapid turns and circling behavior, usually occurring in horizontal planes. You can rotate, to see from any direction, pan, and zoom the 3D chart.
Example 2 - True 3D turn angles show striking differences between treatments.
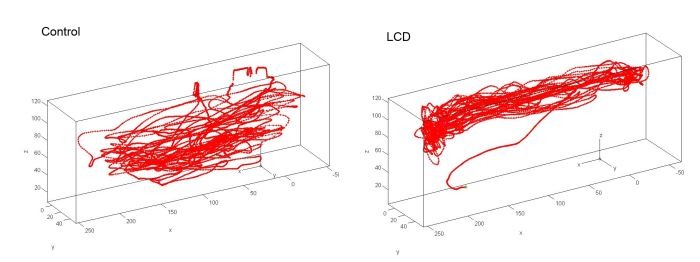
Compared to controls....treatment with LSD evokes a different trajectory in zebrafish
Why you should use 3D tracking
There are several reasons why 3D tracking can offer more than 2D tracking. Here are a few examples:
- Complete trajectory view. In tests like the novel tank, the fish is generally observed from the front side of the tank because bottom diving is one of the most important behaviors to watch out for. However, fish may also exhibit thigmotaxis (swimming close to the walls), which might go unnoticed with a front view camera. 3D tracking allows to you monitor both behaviors and get a clear distinction between them, because you can measure the exact position of the fish at any time.
- Dissecting behaviors. 3D reconstructions of fish trajectories enable macro- and micro-level dissection of quantified behavioral measures in a multi-dimensional, dynamic environment.
- Behavioral phenotyping. Three-dimensional swim path analysis is far more likely to be sensitive to subtle behavior changes brought by experimental treatments (see references below).
Acknowledgements and references
Some of the data on this page was kindly provided by Allan V. Kalueff and his colleagues. For example, see:
- Stewart, A.M.; Grieco, F.; Tegelenbosch, R.A.J.; Kyzar, E.J.; Nguyen, M.; Kaluyeva, A.; Song, C.; Noldus, L.P.J.J.; Kalueff, A.V. (2015). A novel 3D method of locomotor analysis in adult zebrafish: implications for automated detection of CNS drug-evoked phenotypes. Journal of Neuroscience Methods, 255, 66-74.
- Cachat, J.; Stewart, A.; Utterback, E.; Hart, P.; Gaikwad, S.; Wong, K.; Kyzar, E.; Wu, N.; Kalueff, A.V. (2011). Three-dimensional neurophenotyping of adult zebrafish behavior. PLoS ONE, 6(3), e17597.
- Cachat, J.M. (2013). Developing zebrafish models of complex phenotypes relevant to human brain disorders. Neuroscience, Tulane University: New Orleans, 2013: 150.
 English
English German
German French
French Italian
Italian Spanish
Spanish Chinese
Chinese